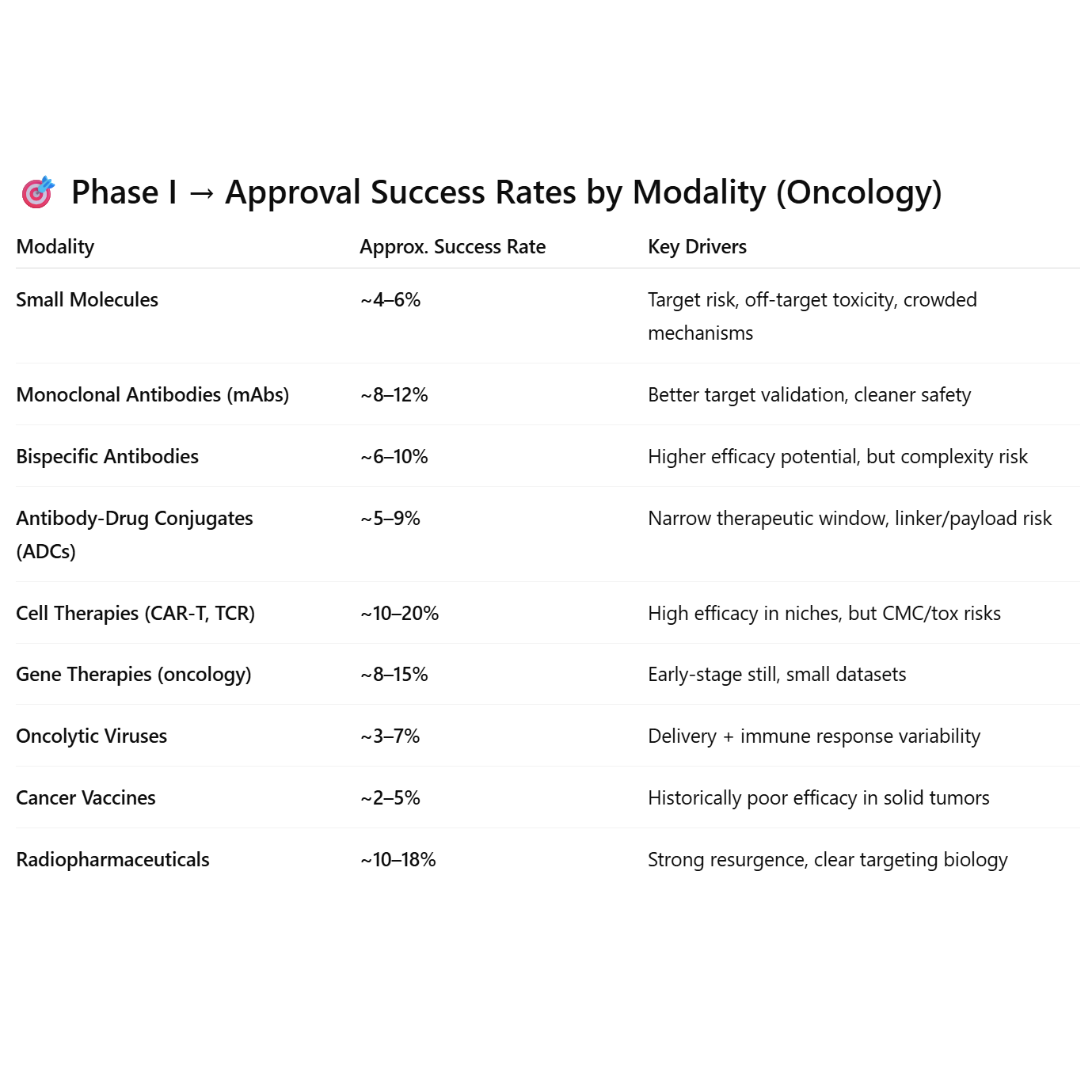
Biology > Modality (but modality is a proxy) for oncology drug success
Modalities with built-in targeting precision (mAbs, radiopharma, CAR-T) outperform. Modalities relying on systemic biology or immune priming (vaccines, viruses) underperform.
~5%–10% of oncology (cancer) drug candidates that enter Phase I ultimately receive U.S. Food and Drug Administration approval. A commonly referenced large-scale analysis (e.g., BIO/industry reports): ~5.1% overall success rate from Phase I → approval for oncology drugs. This is significantly lower than the industry average across all therapeutic areas (~10–15%). Why is oncology lower? Tumor biology is highly complex and heterogeneous. Endpoints (e.g., survival benefit) are harder to demonstrate. Higher toxicity risk tolerance leads to more early-stage entrants but higher attrition. Many programs fail in Phase II due to lack of efficacy. Stage-by-stage (rough ballpark for oncology): Phase I → Phase II: ~60–70%, Phase II → Phase III: ~25–35%, Phase III → Approval: ~50–70%. So, if you’re modeling biotech value or portfolio risk, only ~1 in 10 (and often closer to 1 in 20) oncology drugs entering Phase I will make it to market. But, this number depends a lot on modality. These differences are a byproduct of biology; the modalities operating on complex or interconnected mechanisms are more likely to fail, whereas targeted approaches are more likely to succeed.